Pure bone marrow aspirate injection for chronic Greater Trochanteric Pain Syndrome: a case report
Henderson RG, Colberg RE. Pain Manag. 2018 Jul 1;8(4):271-275 | doi: 10.2217/pmt-2018-0008
Rachel G Henderson and Ricardo E Colberg from Andrews Sports Medicine & Orthopedic Center in Birmingham (AL, USA) show a case of a patient with a debilitating GTPS that was treated with a pure autologous bone marrow aspirate injection into the tendon, and reported near complete resolution of symptoms at the 1-year follow-up.
- Orthobiologic interventions, such as platelet-rich plasma and bone marrow aspirate injections, may stimulate tendon healing and improve pain and function in patients who fail conservative treatment with the current standard of care.
- Given growing concerns with regards to the current bone marrow aspirate concentrate procedure technique, a new technique for aspirating the bone marrow has been introduced.
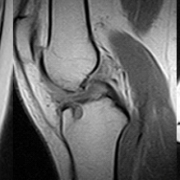
Abstract: There are limited treatment options for patients with a chronic refractory greater trochanteric pain syndrome. Orthobiologic interventions may stimulate tendon healing and improve pain and function in patients who fail the standard conservative treatment. Since the US FDA’s new position statement regarding the centrifugation of bone marrow aspirate products as a potentially ‘more than minimally manipulated’ product, there is a growing concern about the most common bone marrow aspirate concentrate technique. In this case, a 57 year old female with a debilitating chronic greater trochanteric pain syndrome was treated with a pure autologous bone marrow aspirate injection using a novel aspiration technique. The patient showed significant improvements in pain and function without recurrence at 1-year follow-up. This is the first case report to illustrate this novel technique for aspirating pure bone marrow that should comply with the new FDA regulations.
______________________________________________________
The Marrow Cellution™ System is designed to minimize and eliminate the restrictive limitations associated with “point of care” processing systems which employ inefficient traditional aspiration trocar technology and are regulated as drugs “Advanced Therapies Medicinal Products (ATMP)”. Marrow Cellution™ is a minimally invasive bone/cell enriched autograft access and retrieval system, which does not require additional manipulation or processing, it never leaves the sterile field and provides rich autograft without associated morbidity.
Visit our Marrow Cellution product page