EFFICIENT
Never Leaves Sterile Field
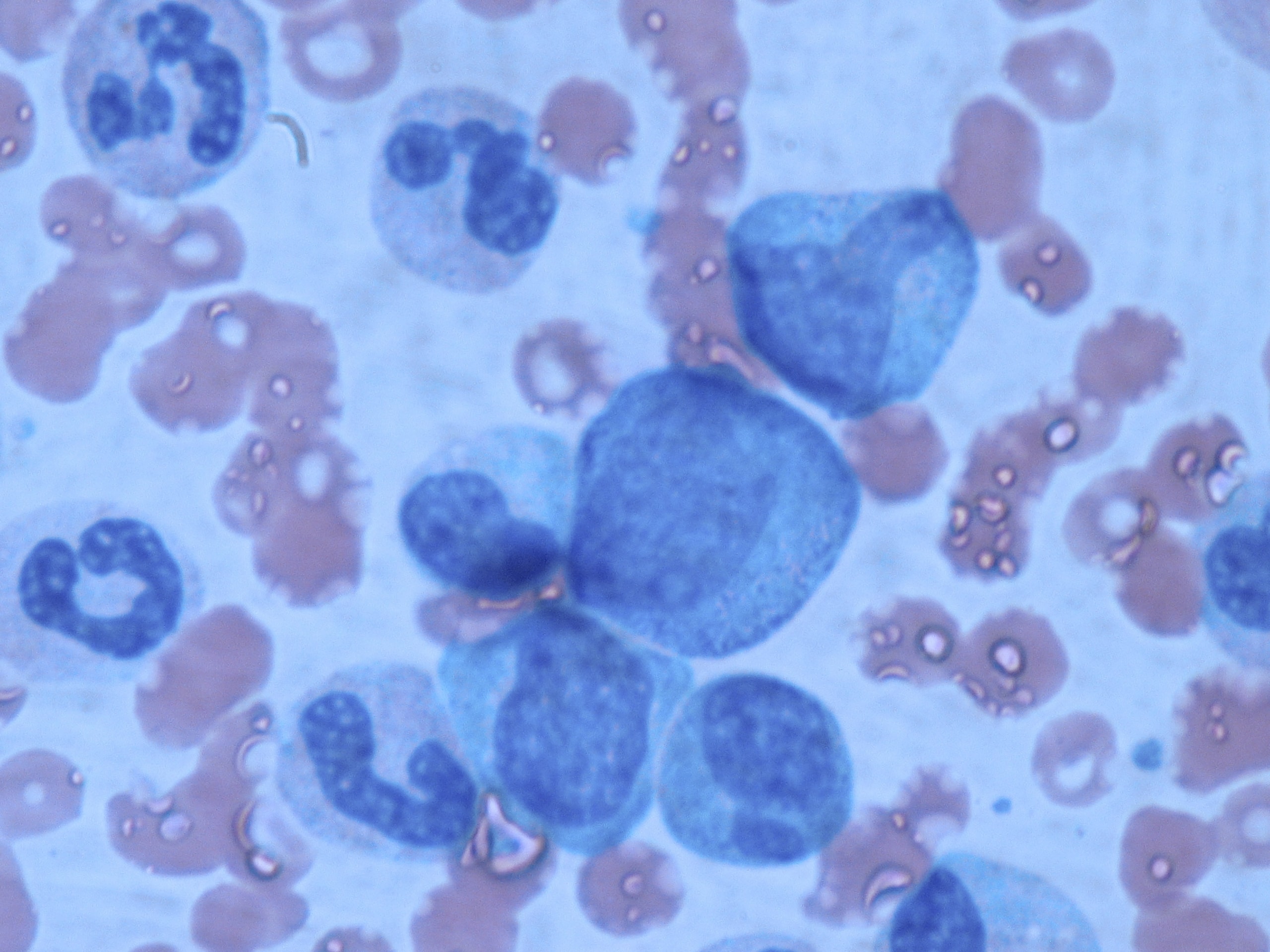
Higher CFU-f Counts per mL
Autologous Bone Graft Collection
Additional Steps Not Required
SPECIFIC
Minimally Invasive
Low Volume—High Yield & Quality
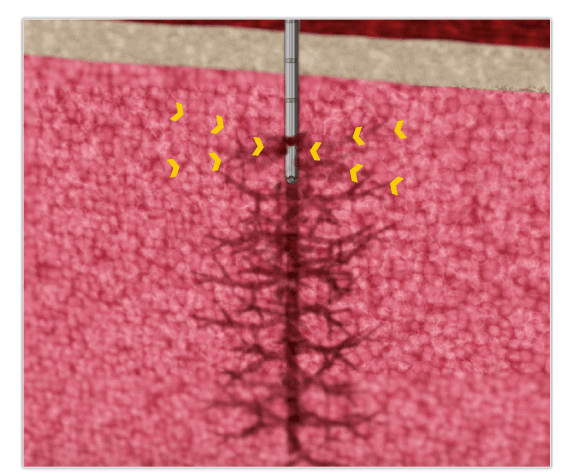
Single Puncture—Multiple Aspirations
Regulatory Compliant
INNOVATIVE
Reduces Excess Blood Collection
Closed-end Aspiration Design
Cannula via Sheath Technology
Lateral vs. Distal Collection