Bone marrow aspirate injection for osteoarthritis of the hip; A pilot study
Tsitsilianos N, Shirazi Z, Lu J, Singh JR.
Interventional Pain Medicine; vol 1, iss 4, 2022, 100163 | doi: 10.1016/j.inpm.2022.100163
Objectives:
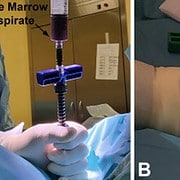
Bone marrow aspirate (BMA) intra-articular injection is a minimally invasive orthobiologic treatment option for osteoarthritis (OA). Hip OA affects a significant portion of the population and has a paucity of data surrounding orthobiologic treatments. The primary objective of this study was to delineate the clinical impact of bone marrow aspirate intra-articular injections on decreasing pain and improving function in patients with hip OA.
Methods:
A single-center, retrospective analysis of thirty-one patients, aged 32 to 83 (62.4 ± 16.5), with Kellgren-Lawrence (KL) Hip OA grading of 2–4 (mean 2.9 ± 0.7), who underwent intra-articular bone marrow aspirate injection into the hip and were followed for twelve months. Evaluation was at baseline, 12 weeks, 6 months, and 12 months using the Numerical Rating Scale (NRS) for pain and the Hip Disability and Osteoarthritis Outcome Score Jr (HOOS-Jr) for function. The proportion of responders, as defined by a ≥50% reduction in NRS pain score, was assessed at 12 weeks, 6 months and 12 months.
Results:
At 6 and 12 months follow-up, there was a statistically significant improvement in NRS scores (P < 0.05). Stratifying by KL grade, subjects with KL grades 2 and 3 experienced statistically significant improvement in NRS scores at 6 and 12 months. Patients with KL grade 4 showed significant improvement in pain at 12 months. Forty-two percent of patients at 6 months and 61% at 12 months reported ≥50% reduction in pain. When stratifying by KL grade, 80% and 71% of KL2 and KL3 grades respectively were responders by 12 months. Patients experienced statistically significant improvement in HOOS-Jr scores at 6 and 12 months.
Conclusion:
In patient with mild, moderate, and severe hip OA, BMA may be an alternative treatment that improves pain and function in patients for as long as 12 months. In addition, BMA may also be an effective, lower cost option to more expensive BMAC preparations.
______________________________________________________
The Marrow Cellution™ System is designed to minimize and eliminate the restrictive limitations associated with “point of care” processing systems which employ inefficient traditional aspiration trocar technology and are regulated as drugs “Advanced Therapies Medicinal Products (ATMP)”. Marrow Cellution™ is a minimally invasive bone/cell enriched autograft access and retrieval system, which does not require additional manipulation or processing, it never leaves the sterile field and provides rich autograft without associated morbidity.
Visit our Marrow Cellution product page