CERVOS BMA – Manual
T-Handle Bone Marrow Aspiration
Ref.: CER-ORTHO-M
The CER-ORTHO-M device incorporates patented technology that limits peripheral blood dilution and eliminates the need for centrifugation.
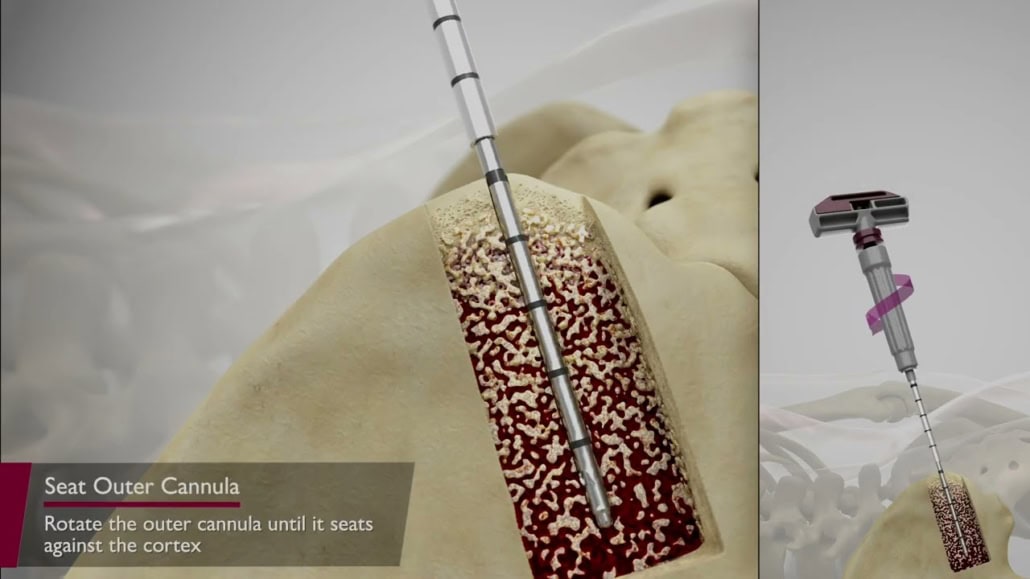
The length makes it ergonomically friendly for accessing the iliac crest and can be advanced wither manually or with a mallet.
The CER-ORTHO-M device uses the patented Marrow Cellution™ technology to limit peripheral blood dilution and eliminate the need for centrifugation.
It accesses aspirate flow collected exclusively laterally as the tip of the aspiration cannula is closed allowing marrow collection perpendicular to and around the entry channel created by the device. It incorporates technology to precisely reposition the retrieval cannula within the marrow space after each aspiration.
This device is often used in conjunction with the CER-BN-84 bone dowel harvesting device and bone graft granules. Combining marrow aspirate with autogenous bone and a graft extender allows clinicians to fill boney defects using the patient’s own cells as the engine for bone healing. The combination of all three provide cells, signals and a scaffold; the building blocks for repairing bone.
Minimize O/R Time
Centrifugation systems typically required 20 minutes or more of spin time during the surgical procedure, not to mention the additional personnel (perfusion) and support time needed for preparation and cleanup of the equipment.
Minimize Sterility Challenges
Centrifugation systems require passing the BMA off the sterile field for processing and back on for implantation. CER-ORTHO-M eliminates the additional steps where infection concerns must be managed.
Minimize Sample Waste
Centrifugation systems typically discard 80% of the aspirate due to the high levels of peripheral blood. Worse, significant numbers of the desired cells (approx. 40%) are discarded because as these cells increase in density prior to division, they are processed into the undesired red cell centrifuge component and thus discarded, substantially limiting the regenerative potential of the resulting sample.
Minimize Use of Anti-Coagulants
Centrifugation systems require at least 10% dilution by volume for the addition of anti-coagulant to allow the sample to separate, and also require another 10% dilution in the form of a neutralizing agent such as thrombin and calcium chloride in order for the marrow to clot in the graft.
Eliminate the Need to Filter
Protocols require the marrow to be filtered prior to centrifugation. Cells bound within a cell aggregate can be delivered to the patient when mixed with graft material or injected. This is not the case when aggregates are filtered out prior to centrifugation. Filtering takes additional time, but more importantly, filtering reduces regenerative potential.
Reduce the Cost of Utilizing Biologics
Delivers a better regenerative solution (more stem cells) at a reduced cost compared to the industry leading centrifugation solutions.
| Article Code: | CER-ORTHO-M |
|---|---|
| Item No.: | 74399-01M |
| Packing: | single sterile packed |